Diapause is an arrested form of life during development, which allows the species to survive extreme stress and then times offspring’s birth to a more favorable environment. In nature, diapause phenomena are widespread throughout the animal kingdom, from simple organisms such as brine shrimps and silkworms, to mammals such as the roe deer, bats, and mice. Our current knowledge of diapause has significantly benefited from the studies in invertebrates. A well-documented example is the C. elegans diapause state called dauer, which helps the species survive a dearth of food. Notably, although organisms in diapause are developmentally suspended, they remain physiologically active and can even be very mobile. For example, C. elegans dauer larvae can move long distances searching for new food sources. In an extreme example, Monarch butterfly in its reproductive diapause form, which holds up the maturation of reproductive organs, can migrate over 3,000 miles from southern Canada and the Midwestern United States to overwintering sites in central Mexico.
Studies of invertebrate diapause have revealed the links between diapause and aging. The individuals that undergo diapause in C. elegans or Drosophila resume a normal lifespan when they reach adulthood. This suggests diapause protects organisms from the damages that accumulate over time. Also, the genetic networks between the C. elegans dauer and aging share many key components, notably the insulin pathway and transcription factor FOXO, suggesting diapause is capable of intervening in aging in invertebrates. However, despite these exciting leads from invertebrates, the knowledge of diapause in vertebrates has remained elusive.
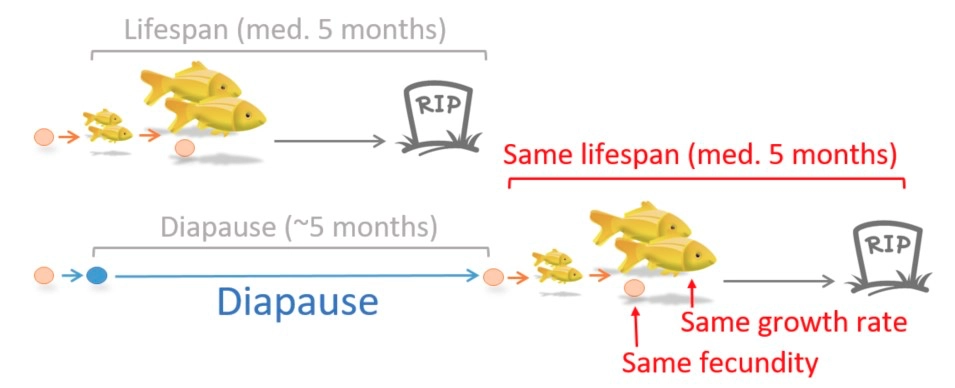
We employed the African killifish (Nothobranchius furzeri ) to study vertebrate diapause to fill this knowledge gap. The African killifish embryos have an intrinsic program to enter and stay in diapause for months to even years. Notably, to hedge the risk, some embryos naturally skip diapause and exhibit a continuous lifecycle (this also allows the African killifish colonies to be maintained in captivity without the hurdle of diapause). Using African killifish’s two natural populations of diapause entering or escaping embryos, and taking advantage of its extremely short lifespan (~5 months) and long diapause (up to years), for the first time, we showed that vertebrates can stay in diapause for a length over their lifespan and still live the subsequent life without tradeoff in growth, fecundity, and lifespan.
Different organisms have diapause occurs at different developmental stages. The African killifish embryos enter diapause at the mid-stage of embryogenesis (between the end of somitogenesis and pharyngula), with ~30,000 cells and many organs already developed (e.g., a segmented brain, a beating heart, muscles in each somite, and clustered germ cells). The digesting track is not yet formed, meaning the endoderm lineage stem cells and their developmental potential are fully preserved throughout the whole diapause. Thus, diapause embryos of the African killifish provide a platform to study the ‘suspended life’ at multiple levels from cells, tissues, organs, to the whole organism.
The developing embryos drastically reprogram their transcriptome to transition into diapause. More than one-third of genes change their expression levels by more than 2 folds in diapause, and the transcriptome no longer resembles a developing organism. Developmental genes (such as key morphogens WNT, FGF, Notch, BMP, Hedgehog) are significantly downregulated and developmental signatures (such as cell proliferation, morphogenesis, differentiation) are globally erased. However, many genes are explicitly upregulated during the transition, maintenance, and exit of diapause, indicating that diapause is an active and dynamic state, with selected genes expressed at different time points for diapause-specific features. The top upregulated genes throughout diapause also include key regulators identified in C. elegans dauer and Drosophila diapause. Therefore, while diapause was independently evolved among organisms, the same genetic toolsets might be repurposed repeatedly via converging evolution. Understand the core program of diapause should help us uncover the genetic and functional principles of dormant biology.